CRISPR is a powerful gene-editing tool that lets scientists change DNA with remarkable precision, speed, and relatively low cost. It began as a natural defense system in bacteria and has now become one of the most important technologies in modern medicine, agriculture, and biotechnology.
What CRISPR Stands For
CRISPR is an acronym for clustered regularly interspaced short palindromic repeats. These are special DNA sequences first discovered in bacteria and archaea, where they act like a genetic memory of past viral infections.
- Each “repeat” is a short DNA sequence that reads the same forward and backward, known as a palindromic repeat.
- Between these repeats sit “spacer” sequences, which come from viruses that previously tried to infect the cell, allowing the cell to recognize those invaders if they return.
Over time, researchers realized these repeats and spacers could be turned into a programmable system for editing genes in many different organisms, not just microbes.
How CRISPR Works In Simple Terms
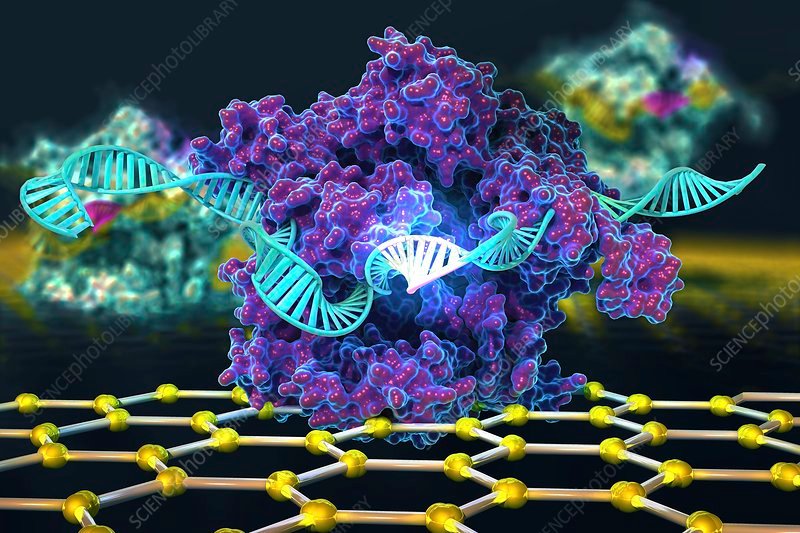
At its core, CRISPR gene editing uses two main components: a guide RNA and a Cas protein that acts like molecular scissors.
- The guide RNA (gRNA or sgRNA) is a short RNA sequence designed to match a specific DNA target inside a cell, effectively telling the system where to go.
- The Cas nuclease, most commonly Cas9, is a protein that binds to the guide RNA and then cuts the DNA at the chosen site.
When Cas9 and the guide RNA come together, they form a ribonucleoprotein complex that scans DNA for a short sequence called a PAM (protospacer adjacent motif). Once the complex finds a matching DNA sequence next to a PAM, Cas9 creates a precise break in both DNA strands, giving cells an opportunity to repair the break and, in the process, add, remove, or replace genetic material.
What Happens After DNA Is Cut
The way a cell repairs the cut largely determines the final edit. Cells mainly rely on two natural repair pathways to fix the double-strand break created by CRISPR.
- Non-homologous end joining (NHEJ) quickly seals the break but often introduces small insertions or deletions, which can disrupt a gene and effectively “knock it out.”
- Homology-directed repair (HDR) uses a DNA template provided by researchers to insert new sequences or correct mutations with much higher accuracy, although this pathway is usually less efficient.
Newer CRISPR variants, including base editors and prime editors, avoid fully cutting both DNA strands and instead change individual letters or short stretches of DNA with fewer unwanted side effects.
Real-World Uses Of CRISPR Today
CRISPR has moved quickly from lab benches into real clinical and industrial applications. In human health, it is being tested or used to treat inherited blood disorders, eye diseases, and some immune-related conditions.
- Clinical research is exploring how CRISPR can correct harmful mutations in blood-forming cells, with the goal of offering long-term relief or even a potential cure for some genetic diseases.
- In agriculture, scientists are using CRISPR to develop crops that tolerate drought, resist pests, or offer enhanced nutrition, often faster and more precisely than conventional breeding.
Researchers are also exploring CRISPR-based tools for controlling disease-carrying insects, editing microbes for cleaner manufacturing processes, and designing new therapies that edit RNA instead of DNA.
Can You Use Cre-Lox With CRISPR?
Cre-lox and CRISPR are two different genetic tools, and they can be combined in the same experimental system. Cre-lox uses an enzyme called Cre recombinase and short DNA sites called loxP to rearrange or delete DNA segments without making double-strand breaks in the same way CRISPR does.
- Some advanced bacterial genome engineering strategies insert a “landing pad” containing multiple lox sites and then add CRISPR-Cas components using Cre-lox–mediated cassette exchange, allowing precise integration and later clean removal of editing machinery.
- This kind of combined design lets scientists take advantage of the flexibility of Cre-lox for DNA rearrangements and the targeting precision of CRISPR-Cas for specific edits in the same cell or organism.
Because the two systems operate through distinct enzymes and recognition sites, researchers can plan workflows where Cre-lox controls when and where CRISPR components are active, or vice versa, giving much finer control over complex genome engineering projects.
Why CRISPR Matters For The Future
CRISPR is reshaping how experts think about treating disease, improving food security, and understanding biology at a fundamental level. Its relative simplicity—designing a new guide RNA is often enough to retarget the system—means that more laboratories can participate in gene-editing research than ever before.
At the same time, the power of this technology raises important ethical and regulatory questions, including how to ensure safety, fairness, and responsible use as clinical applications expand. Ongoing work focuses on improving accuracy, reducing off-target effects, and building versions of CRISPR that can be turned on or off with higher precision, making future applications both safer and more effective.